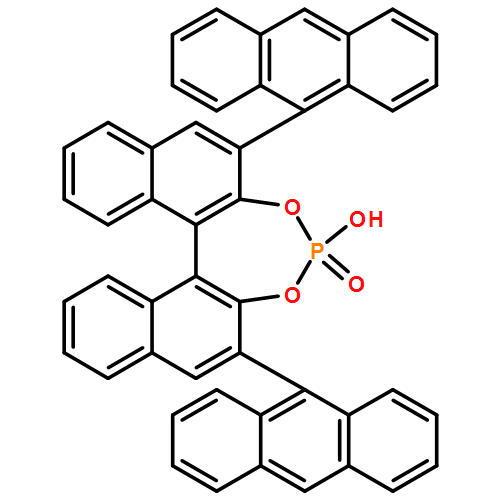
The condensation cyclization between isatins and 5-methoxy tryptamine catalyzed by chiral phosphoric acids provides spirooxindole tetrahydro-β-carboline products in excellent yields (up to 99%) and enantioselectivity (up to 98:2 er). A comparison of catalysts provides insight for the substrate scope and factors responsible for efficient catalytic activity and selectivity in the spirocyclization. Chiral phosphoric acids with different 3,3′-substitution on the binaphthyl system and opposite axial chirality afford the spiroindolone product with the same absolute configuration.