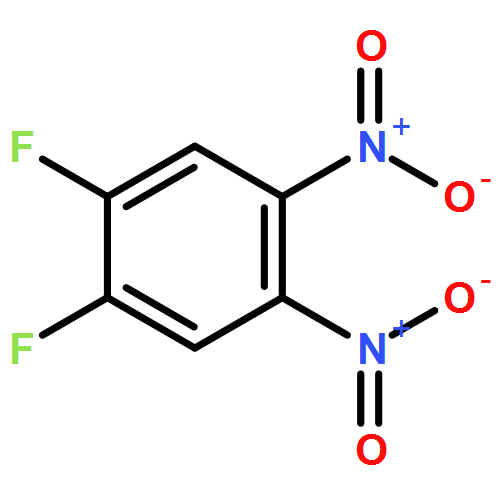
•Two novel donor-π-acceptor type of conjugated polymers were synthesized.•Selenophene copolymer showed higher absorptivity and intermolecular ordering.•Device performances improved obviously after replacing thiophene with selenophene.Two novel donor-π-acceptor type of conjugated polymers with thiophene or selenophene as the π-bridge between the electron-donating indolocarbazole and electron-accepting difluorobenzotriazole unit, were designed and synthesized through a palladium catalyzed Suzuki polymerization. The replacement of the thiophene bridge by selenophene shows negligible effects on the geometries of molecular chain, as indicated by theoretical calculation on the basis of density of functional theory. However, obvious bathochromic shift along with the increased intensity in the UV–vis absorption profile of selenophene based copolymers relative to their thiophene based counterparts are realized, as can be attributed to the more pronounced heavy atom effects of the selenium than the sulfur atom. Compared to the thiophene based copolymer, the selenophene based copolymer exhibited about one order of magnitude increase in hole mobility, from 0.0014 to 0.01 cm2 V−1 s−1, and about two times of magnitude increase in power conversion efficiency, from 1.01% to 2.39%, as evaluated by field effect transistors and bulk heterojunction polymer solar cells, respectively. These results indicated that the selection of appropriate π-bridge by is crucial for the improvement of performance of π-conjugated polymers.