Co-reporter: Mingli Deng, Feilong Yang, Pan Yang, Zhouxun Li, Jinyu Sun, Yongtai Yang, Zhenxia Chen, Linhong Weng, Yun Ling, and Yaming Zhou
pp: 5794
Publication Date(Web):October 27, 2015
DOI: 10.1021/acs.cgd.5b01155
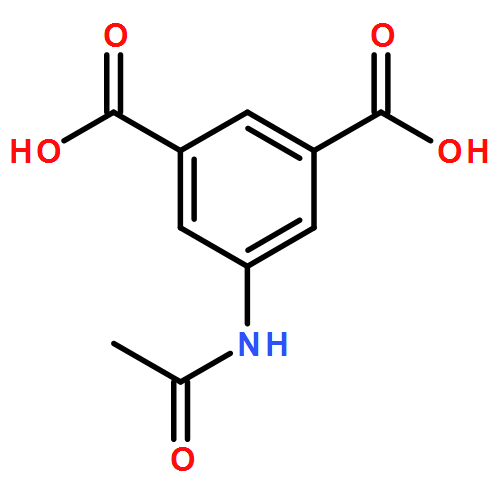
Metal–organic frameworks with tunable functional groups like solid-solution have received considerable interest. In this paper, a crs-type structure built of [Zn3(OH)(dmtrz)3] triazolate-trinuclear and [Zn2(COO)4] paddlewheel units, linked by 3,5-dimethyl-1,2,4-triazolate (dmtrz) and isophthalate (ipa) ligands (named as MAC-4-A), has been used as a prototype framework. Four types of functional groups (B: 5-hydroxyisophthalate, C: 5-aminoisophthalate, D: 5-ethoxyisophthalate, and E: 5-acetamidoisophthalate) have been integrated into the framework, giving isostructures of MAC-4-B to E, respectively. Then, by the solid-solution framework approach, four group samples are prepared, which are MAC-4-AB-x, MAC-4-AC-x, MAC-4-AD-x, and MAC-4-AE-x (x denoted as the ratio of functional ligands in the framework, x = 0.3, 0.5, 0.7), and their CO2 and N2 adsorptions have been studied. Our results revealed that the CO2 capacity is enhanced with the increase of functional groups and then decreases, showing a maximum uptake amount on MAC-4-AB-0.5, MAC-4-AC-0.5, MAC-4-AD-0.7, and MAC-4-AE-0.5 at 298 K and 1 bar, respectively. On the other hand, the calculated selectivity of CO2 over N2 gradually increases, giving the highest selectivity after the pore surface is completely functionalized.