Co-reporter: Kou-Lin Zhang, Chuan-Tao Hou, Jing-Jing Song, Ye Deng, Liang Li, Seik Weng Ng and Guo-Wang Diao
pp: 590-600
Publication Date(Web):07 Nov 2011
DOI: 10.1039/C1CE05577A
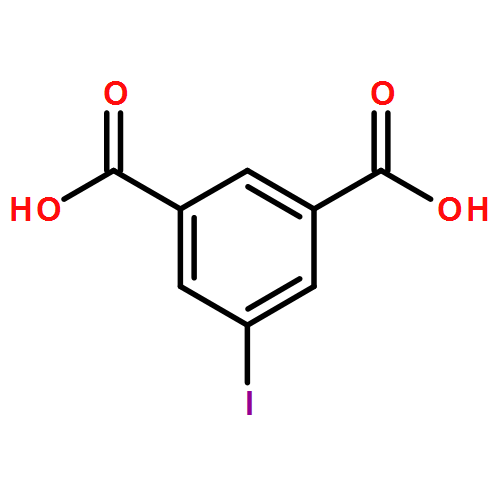
Two temperature-dependent structures of 2D and 3D Zn(II)-organic frameworks (ZOFs) with a new 5-substituted benzene-1, 3-dicarboxylic ligand, 5-iodoisophthalic acid (H2IIP), and an auxiliary flexible ligand, 1,4-bis(1,2,4-triazol-1-yl)butane (btb), with different motifs, have been investigated. Results show that when the reaction was carried out at room temperature, a undulating 2D (4,4)-network, {[Zn(IIP)(btb)]·4H2O}n (1), which further extends into a novel “soft” 3D supramolecular microporous framework with two kinds of 1D nanochannels supported by face to face π⋯π stacking interactions and C–I⋯I halogen bonds, was generated. Under hydrothermal condition at 170 °C, however, a two-fold interpenetrated 3D framework with α-Po network topology, [Zn(IIP)(btb)]n (2), would be obtained. Interestingly, both the right- and left-handed 21 helical water chains lie in one kind of the nanochannels in 1. When the auxiliary ligand was replaced by a less flexible one with a shorter spacer length, 1,3-bis(1,2,4-triazol-1-yl)propane (btp), a novel temperature-independent single-walled discrete coordination tube, {[Zn(IIP)(btp)]·2H2O}n (3), was obtained at the same two temperatures. Inside the tube is found the 21 helical water chain. Interestingly, the reversible desorption/adsorption behavior to water is significantly observed in the frameworks 1 and 3. The framework 1 falls within the category of “recoverable collapsing” and “guest-induced re-formation” frameworks. The result shows their potential application as late-model water absorbents in the field of adsorption materials. Remarkably, the first discrete single-walled Zn(II) coordination tube 3 shows high framework stability and exhibits reversible desorption/adsorption to some small guest organic molecules (methanol, ethanol and isopropanol). Furthermore, these compounds exhibit blue fluorescence in the solid state.