Co-reporter: Renee Bouley; Derong Ding; Zhihong Peng; Maria Bastian; Elena Lastochkin; Wei Song; Mark A. Suckow; Valerie A. Schroeder; William R. Wolter; Shahriar Mobashery;Mayland Chang
pp: 5011-5021
Publication Date(Web):April 18, 2016
DOI: 10.1021/acs.jmedchem.6b00372
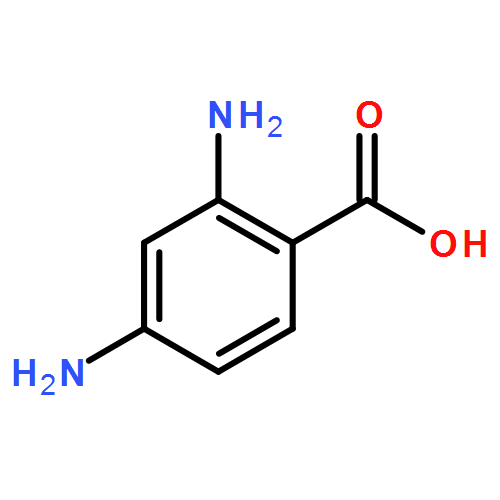
We recently reported on the discovery of a novel antibacterial (2) with a 4(3H)-quinazolinone core. This discovery was made by in silico screening of 1.2 million compounds for binding to a penicillin-binding protein and the subsequent demonstration of antibacterial activity against Staphylococcus aureus. The first structure–activity relationship for this antibacterial scaffold is explored in this report with evaluation of 77 variants of the structural class. Eleven promising compounds were further evaluated for in vitro toxicity, pharmacokinetics, and efficacy in a mouse peritonitis model of infection, which led to the discovery of compound 27. This new quinazolinone has potent activity against methicillin-resistant (MRSA) strains, low clearance, oral bioavailability and shows efficacy in a mouse neutropenic thigh infection model.