Co-reporter: Konstantin V. Luzyanin ; Pavel V. Gushchin ; Armando J. L. Pombeiro ; Matti Haukka ; Victor I. Ovcharenko ;Vadim Yu. Kukushkin
pp: 6919-6930
Publication Date(Web):June 27, 2008
DOI: 10.1021/ic800481a
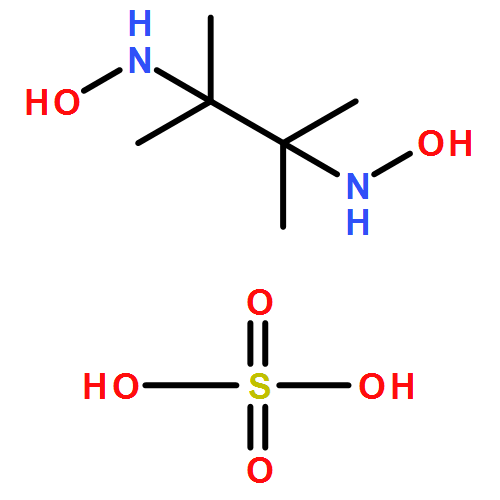
The reaction of K2[PtCl4] and HO(H)NCMe2CMe2N(H)OH·H2SO4 (BHA·H2SO4; 2) in a molar ratio 1:2 at 20−25 °C in water affords a mixture of [Pt(BHA)2][PtCl4] (5) and [Pt(BHA-H)2] (6) (BHA-H = anionic monodeprotonated form of BHA) which, upon heating at 80−85 °C for 12 h or on prolonged keeping at 20−25 °C for 2 weeks, is subject to a slow transformation giving [PtCl2(BHA)] (7). The latter compound is also obtained from the reaction between K[PtCl3(Me2SO)] and 2. The chlorination of [PtCl2(BHA)] (7) in freshly distilled dry chloroform leads to the selective oxidation of one N(H)OH group yielding [PtCl2{HO(H)NCMe2CMe2N=O}] (13), while the chlorination in water produces the complex [PtCl2(O=NCMe2CMe2N=O)] (14) bearing the unexplored dinitrosoalkane species. Treatment of 14 with 2 equiv of 1,2-bis-(diphenylphosphino)ethane (dppe) in CH2Cl2 results in the liberation of the dinitrosoalkane ligand followed by its fast cyclization giving the α-dinitrone (3,3,4,4-tetramethyl-1,2-diazete-1,2-dioxide) in solution and the solid [Pt(dppe)2](Cl)2. The PtII complexes with hydroxylamino∩oximes [PtCl2{HO(H)NC(Me)2C(R)=NOH}] (R = Me 8; R = Ph 9) upon their oxidation with Cl2 in CHCl3 afford the nitrosoalkane derivatives [PtCl2{O=NCMe2C(R)=NOH}] (R = Me 16; Ph 17), respectively, while the corresponding chlorination of the bis-chelates [Pt{HO(H)NCMe2C(R)=NOH}2] (R = Me 10; Ph 11) gives [Pt{O=NCMe2C(R)=NO}2] (R = Me 18; Ph 19). The formulation of 5−19 is based on C, H, and N microanalyses, IR, 1D (1H, 13C{1H}, 195Pt) and 2D (1H,1H-COSY,1H,13C-HSQC) NMR spectroscopies, and X-ray diffraction for five complexes (5, 7, and 12−14).