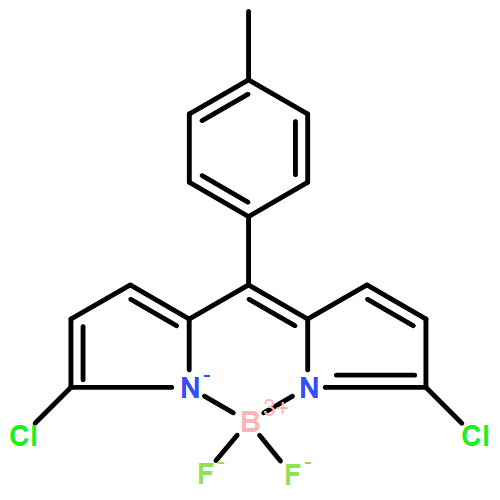
Simple organic molecules (SOM) based on bis(haloBODIPY) are shown to enable circularly polarized luminescence (CPL), giving rise to a new structural design for technologically valuable CPL-SOMs. The established design comprises together synthetic accessibility, labile helicity, possibility of reversing the handedness of the circularly polarized emission, and reactive functional groups, making it unique and attractive as advantageous platform for the development of smart CPL-SOMs.