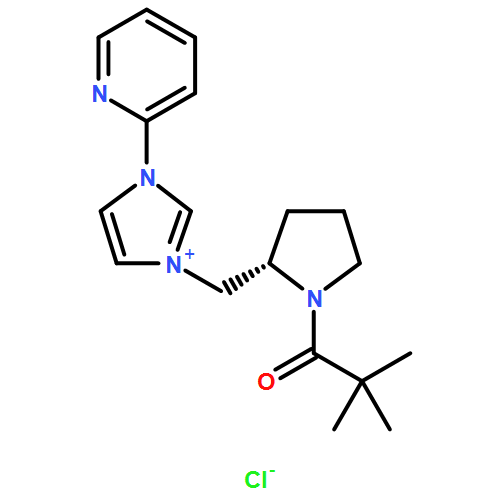
Abstract
Amino acid-derived chiral imidazolium salts, each bearing a pyridine ring, were developed as N-heterocyclic carbene ligands. The copper-catalyzed asymmetric alkylation of various N-sulfonylimines with dialkylzinc reagents in the presence of these chiral imidazolium salts afforded the corresponding alkylated products with high enantioselectivity (up to 99 % ee). The addition of HMPA to the reaction mixture as a co-solvent is critical in terms of chemical yield and enantioselectivity. A wide range of N-sulfonylimines and dialkylzinc reagents were found to be applicable to this reaction.