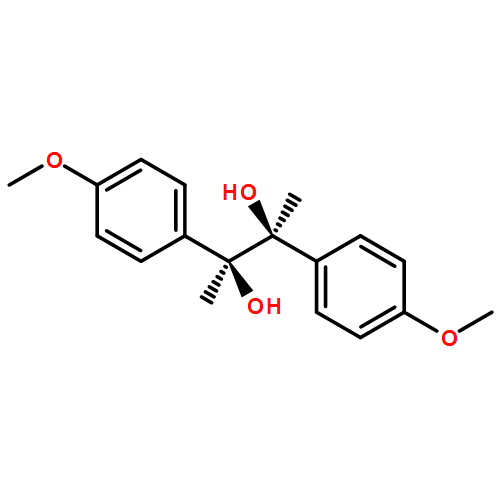
Novel diastereoselective photo-induced pinacol coupling reactions of acetophenones by using triethylamine and chiral tertiary amines as electron donating co-sensitizers were studied. Various influence factors including solvents, substituents, and chiral amines on both the diastereoselectivity and yield were examined. The diastereoselectivities were enhanced in supramolecular systems of cyclodextrins and zeolites. The best result of dl/meso up to 82:18 was obtained in combination use of chiral tertiary amine and β-CD.A novel diastereoselective photo-induced pinacol coupling reactions of substituted acetophenones by electron transfer photosensitization using triethylamine and chiral tertiary amines as electron donating co-sensitizers were investigated in β-CD.