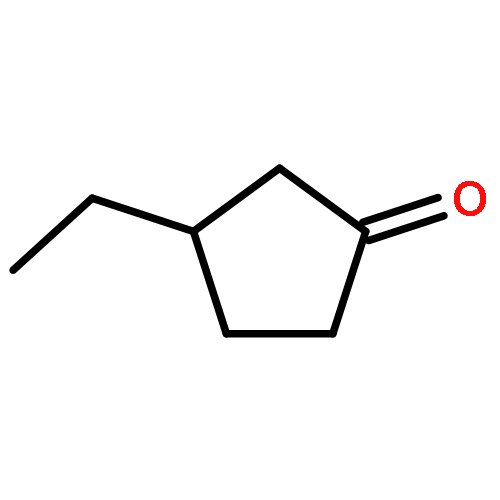
A library of chiral phosphine–phosphite ligands was evaluated in the Cu-catalyzed asymmetric 1,4-addition of Grignard reagents to cyclopentenone, cycloheptenone and 5,6-dihydro-2H-pyran-2-one. TADDOL-based ligands 1a and 1b with a bulky substituent at the ortho-position to the chiral phosphite moiety gave rise to the 1,4-addition products with high enantioselectivities (up to 93% ee). In addition to ethyl-MgBr (as a standard alkyl nucleophile) phenyl- and isopropenyl-MgBr could also be employed. In the case of cyclopentenone, the use of chlorotrimethylsilane as an additive led to improved regio- and enantioselectivities.


3-Ethyl-cyclopentanoneC7H12O93% ee

[α]58925=+90 (c 1, CHCl3)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (R)

(R)-3-(2-Propenyl)-cyclopentanoneC8H12O89% ee

[α]58920=+87.8 (c 0.32, CHCl3)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (R)

(R)-3-Phenyl-cyclopentanoneC12H12O57% ee

[α]58920=+47.2 (c 1, CH2Cl2)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (R)

(S)-3-Ethyl-cycloheptanoneC9H16O91% ee

[α]58920=+62.1 (c 1.1, CH3CN)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (S)

(R)-3-(2-Propenyl)-cycloheptanoneC10H16O86% ee

[α]58920=+68.2 (c 1.1, CH2Cl2)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (R)

(R)-3-Phenyl-cycloheptanoneC13H16O90% ee

[α]58920=+54.2 (c 0.9, CH2Cl2)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (R)

(S)-4-Ethyl-tetrahydro-pyrane-2-oneC7H12O2

[α]58920=-16.5 (c 0.95, CHCl3)Source of chirality: enantioselective catalysis: (R,R)-taddolAbsolute configuration: (S)