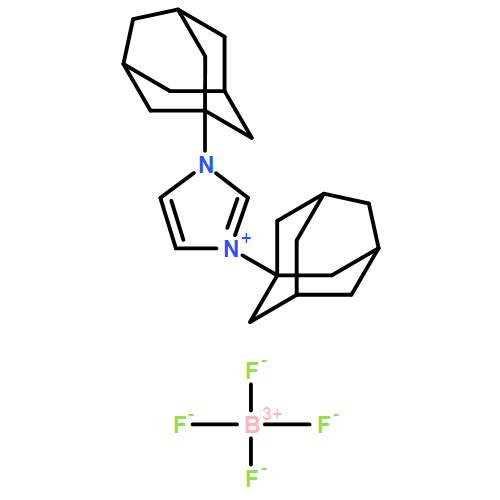
BASIC PARAMETERS Find an error
Multicomponent catalytic enantioselective transformations that entail the combination of butadiene or isoprene (common feedstock), an enoate (prepared in one step) and B2(pin)2 (commercially available) are presented. These processes constitute an uncommon instance of conjugate addition of an allyl moiety and afford the desired products in up to 83 % yield and 98:2 enantiomeric ratio. Based on DFT calculations stereochemical models and rationale for the observed profiles in selectivity are provided.