Co-reporter: Fangting Yu, Vincent L. Pecoraro
pp: 99-105
Publication Date(Web):12 November 2013
DOI: 10.1016/j.poly.2013.02.074
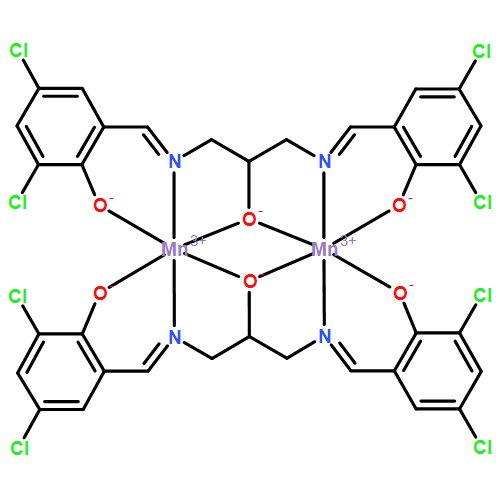
•First direct evidence of a manganyl species in the mixed-valent [Mn(2-OH Salpn)]2 series.•The use of a mechanistic probe to demonstrate the formation of a fleeting intermediate MnIVMnVO.•Directly relevant to the chemistry that might occur during photosynthetic water oxidation.High-valent Mn species are key intermediates in the oxygen evolving complex in photosystem II. In some mechanisms, the MnVO moiety is proposed to be a critical species at the final step of oxygen production. This work reports the use of a mechanistic probe 2-methyl-1-phenylpropan-2-yl hydroperoxide (MPPH) to distinguish the formation of MnIVMnIV(OH) and MnIVMnVO species. We demonstrate that a dimeric Mn complex [MnIIIMnIV(2-OH-3,5-diClsalpn)]+ reacts with MPPH, leading to the production of the O–O bond heterolysis product. In this process, the Mn complex transforms into a transient MnIVMnVO species, which undergoes comproportionation rapidly.We use a mechanistic probe 2-methyl-1-phenylpropan-2-yl hydroperoxide (MPPH) to investigate the formation of a transient manganyl species in a dimeric Mn complex, which is a synthetic model for the oxygen evolving complex in photosystem II. We have obtained the first direct evidence of a manganyl species in the mixed-valent [Mn(2-OH Salpn)]2 series.Download full-size image