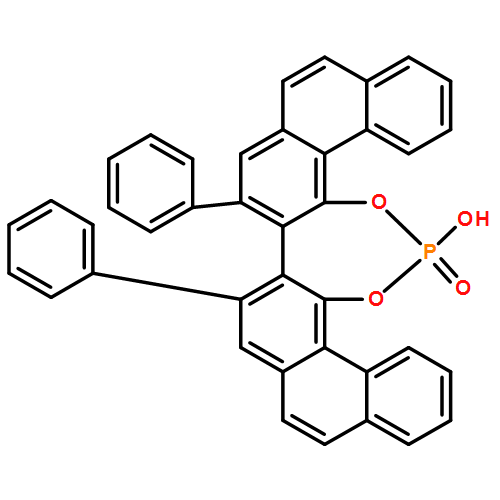
Co-reporter: Kazuhiro Higuchi, Shin Suzuki, Reeko Ueda, Norifumi Oshima, Emiko Kobayashi, Masanori Tayu, and Tomomi Kawasaki
pp: 154-157
Publication Date(Web):December 19, 2014
DOI: 10.1021/ol5033865
The asymmetric total synthesis of (−)-leuconoxine has been achieved. The desymmetrization of a prochiral diester using a chiral phosphoric acid catalyst produced a highly enantioenriched lactam with excellent yield. The ring construction featuring an intramolecular N-acyliminium cyclization and the one-step pyrrolidone formation using Bestmann’s ylide was successfully accomplished.