BASIC PARAMETERS Find an error
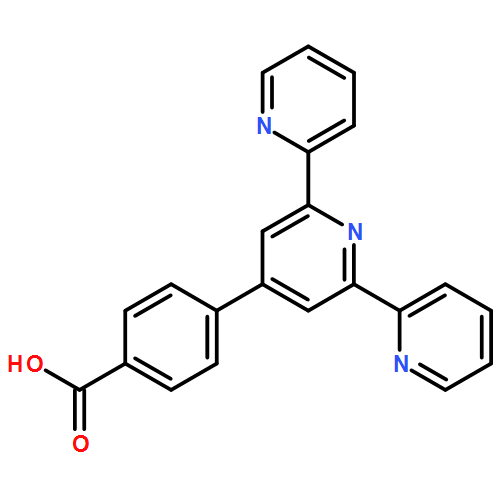
Linkers that favor rectification of interfacial electron transfer are likely to be required for efficient photo-driven catalysis of multi-electron reactions at electrode surfaces. Design principles are discussed, together with the synthesis and characterization of a specific pair of molecular linkers, related by inversion of the direction of an amide bond in the heart of the molecule. The linkers have a terpyridyl group that can covalently bind Mn as in a well-known water oxidation catalyst and an acetylacetonate group that allows attachment to TiO2 surfaces. The appropriate choice of the sense of the amide linkage yields directionality of interfacial electron transfer, essential to enhance electron injection and slow back-electron transfer. Support comes from electron paramagnetic resonance and terahertz spectroscopic measurements, as well as computational modeling characterizing the asymmetry of electron transfer properties.
Linkers that favor rectification of interfacial electron transfer are likely to be required for efficient photo-driven catalysis of multi-electron reactions at electrode surfaces. Design principles are discussed, together with the synthesis and characterization of a specific pair of molecular linkers, related by inversion of the direction of an amide bond in the heart of the molecule. The linkers have a terpyridyl group that can covalently bind Mn as in a well-known water oxidation catalyst and an acetylacetonate group that allows attachment to TiO2 surfaces. The appropriate choice of the sense of the amide linkage yields directionality of interfacial electron transfer, essential to enhance electron injection and slow back-electron transfer. Support comes from electron paramagnetic resonance and terahertz spectroscopic measurements, as well as computational modeling characterizing the asymmetry of electron transfer properties.
Linkers that favor rectification of interfacial electron transfer are likely to be required for efficient photo-driven catalysis of multi-electron reactions at electrode surfaces. Design principles are discussed, together with the synthesis and characterization of a specific pair of molecular linkers, related by inversion of the direction of an amide bond in the heart of the molecule. The linkers have a terpyridyl group that can covalently bind Mn as in a well-known water oxidation catalyst and an acetylacetonate group that allows attachment to TiO2 surfaces. The appropriate choice of the sense of the amide linkage yields directionality of interfacial electron transfer, essential to enhance electron injection and slow back-electron transfer. Support comes from electron paramagnetic resonance and terahertz spectroscopic measurements, as well as computational modeling characterizing the asymmetry of electron transfer properties.
Linkers that favor rectification of interfacial electron transfer are likely to be required for efficient photo-driven catalysis of multi-electron reactions at electrode surfaces. Design principles are discussed, together with the synthesis and characterization of a specific pair of molecular linkers, related by inversion of the direction of an amide bond in the heart of the molecule. The linkers have a terpyridyl group that can covalently bind Mn as in a well-known water oxidation catalyst and an acetylacetonate group that allows attachment to TiO2 surfaces. The appropriate choice of the sense of the amide linkage yields directionality of interfacial electron transfer, essential to enhance electron injection and slow back-electron transfer. Support comes from electron paramagnetic resonance and terahertz spectroscopic measurements, as well as computational modeling characterizing the asymmetry of electron transfer properties.