Co-reporter:Ahmed A. Zaki, Zulfiqar Ali, Yan-Hong Wang, Yasser A. El-Amier, Shabana I. Khan, Ikhlas A. Khan
Steroids 2017 Volume 125(Volume 125) pp:
Publication Date(Web):1 September 2017
DOI:10.1016/j.steroids.2017.06.003
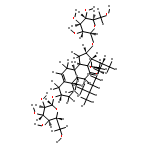
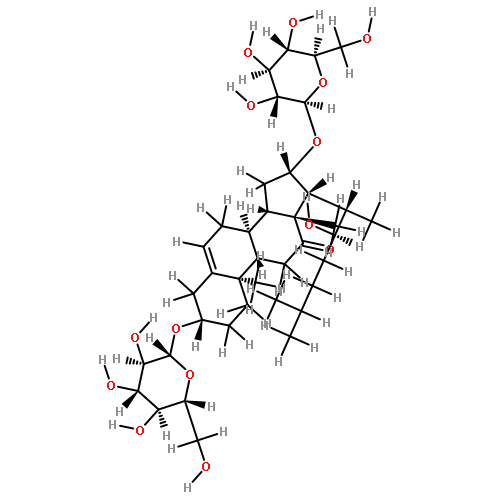
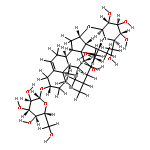
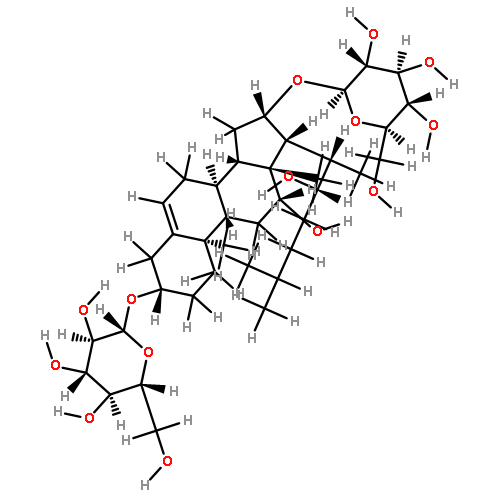
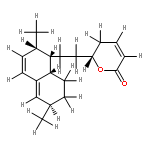
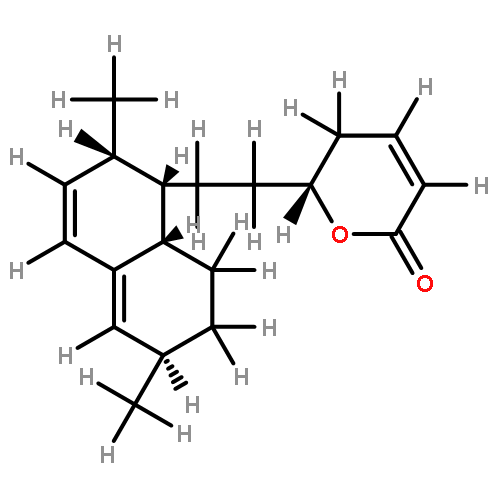
•Seven steroidal saponins were isolated from Panicum turgidum.•Three bidesmosidic cholestane-type and one spirostane-type steroidal glycosides were found new.•Spirostane-type steroidal glycosides showed cytotoxicity.Three new bidesmosidic cholestane-type steroidal glycosides, 16-O-β-d-glucopyranosyl-cholest-5-en-3β,16β-diol-22-one-3-O-α-l-rhamnopyranosyl-(1 → 2)-O-[(β-d-glucopyranosyl(1 → 4)]-O-β-d-glucopyranoside (1), 16-O-β-d-glucopyranosylcholest-5-en-3β,16β-diol-22-one-3-O-α-l-rhamnopyranosyl-(1 → 2)-O-β-d-glucopyranoside (2), and 16-O-β-d-glucopyranosylcholestan-3β,16β-diol-6,22-dione-3-O-α-l-rhamnopyranosyl-(1 → 2)-O-β-d-glucopyranoside (3) were isolated from a methanolic extract of Panicum turgidum. In addition four known compounds, pennogenin 3β-O-α-l-rhamnopyranosyl-(1 → 2)-O-[α-l-rhamnopyranosyl-(1 → 4)-O-α-l-rhamnopyranosyl-(1 → 4)]-O-β-d-glucopyranoside (4), yamogenin 3β-O-α-l-rhamnopyranosyl-(1 → 2)-O-[α-l-rhamnopyranosyl-(1 → 4)]-O-β-d-glucopyranoside (5), yamogenin 3β-O-α-l-rhamnopyranosyl-(1 → 2)-O-[α-l-rhamnopyranosyl-(1 → 4)-O-α-l-rhamnopyranosyl-(1 → 4)]-O-β-d-glucopyranoside (6), and pennogenin 3β-O-α-l-rhamnopyranosyl-(1 → 2)-O-[α-l-rhamnopyranosyl-(1 → 4)]-O-β-d-glucopyranoside (7) were also isolated and characterized. Their structures were established using extensive spectroscopic methods including 1D and 2D NMR and HRESIMS. The isolated compounds were screened for cytotoxicity towards a panel of mammalian cell lines and 4–7 were found to be cytotoxic.Download high-res image (130KB)Download full-size image
Co-reporter:Zulfiqar Ali, Hatice Demiray, Ikhlas A. Khan
Tetrahedron Letters 2014 Volume 55(Issue 2) pp:369-372
Publication Date(Web):8 January 2014
DOI:10.1016/j.tetlet.2013.11.031
A new indole alkaloid, 7β-hydroxy-7H-mitraciliatine (1) and a new oxindole alkaloid, isospeciofoleine (2) together with nine known alkaloids were isolated from Mitragyna speciosa and characterized by NMR, CD, and MS spectroscopic data analyses. The 1H and 13C NMR spectroscopic data of isospeciofoline (3), isorotundifoline (4), paynantheine (5), and 3-isopaynantheine (6) were also reported for the first time.
Co-reporter:Ramsay S. T. Kamdem, Pascal Wafo, Sammer Yousuf, Zulfiqar Ali, Achyut Adhikari, Saima Rasheed, Ikhlas A. Khan, Bonaventure T. Ngadjui, Hoong-Kun Fun, and M. Iqbal Choudhary
Organic Letters 2011 Volume 13(Issue 20) pp:5492-5495
Publication Date(Web):September 30, 2011
DOI:10.1021/ol202217d
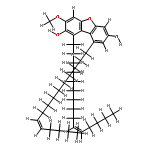
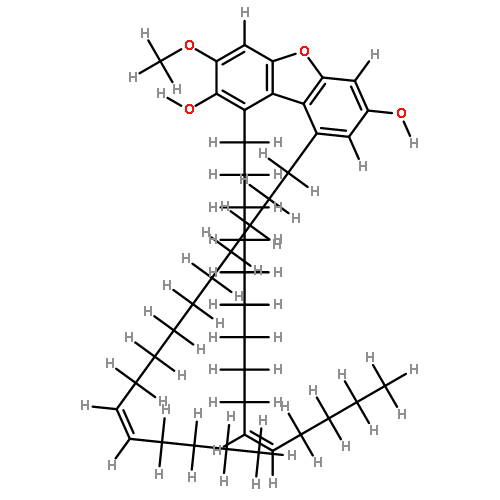
Canarene (1), a triterpene with an unprecedented carbon backbone, was isolated from Canarium schweinfurthii. It is the first member of a new class of triterpenoids, for which the name “canarane’’ is proposed. 1 showed weak α-glucosidase inhibitory activity, and its structure was unambiguously deduced by single-crystal X-ray diffraction.