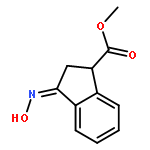
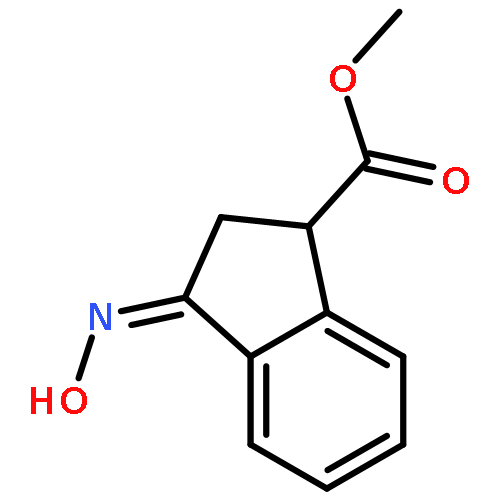
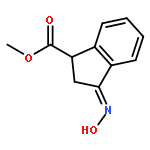
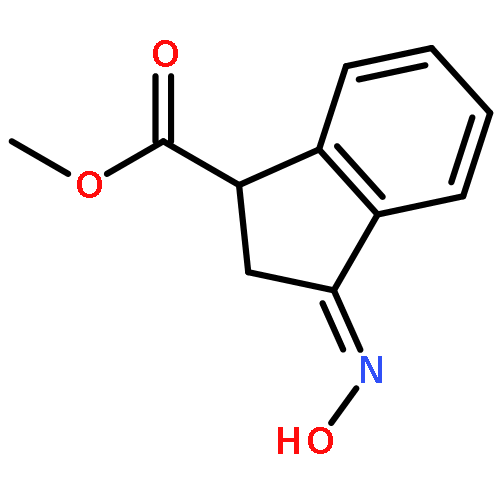
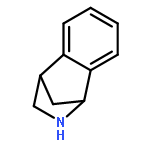
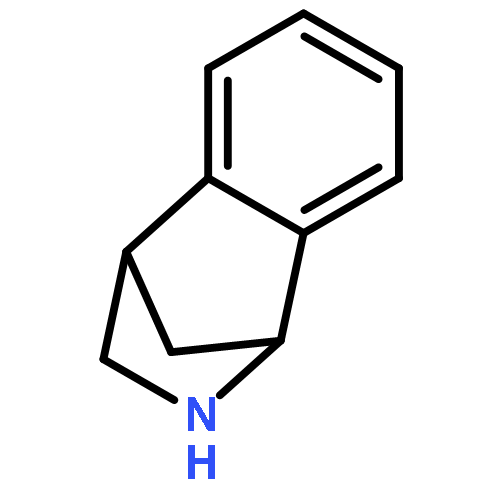
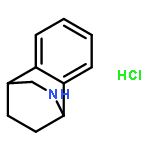
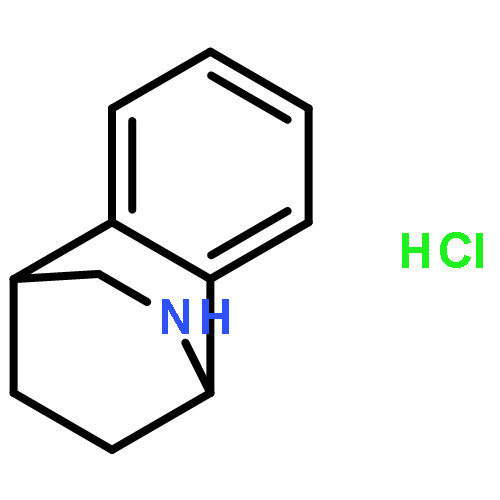
Co-reporter:Gary L. Grunewald, F. Anthony Romero, Mitchell R. Seim, Kevin R. Criscione, Jean D. Deupree, Christy C. Spackman, David B. Bylund
Bioorganic & Medicinal Chemistry Letters 2005 Volume 15(Issue 4) pp:1143-1147
Publication Date(Web):15 February 2005
DOI:10.1016/j.bmcl.2004.12.013
3-Hydroxyethyl- and 3-hydroxypropyl-7-substituted-tetrahydroisoquinolines (9, 10, 16, and 17) were synthesized and evaluated for their phenylethanolamine N-methyltransferase (PNMT) inhibitory potency and affinity for the α2-adrenoceptor. Although α2-adrenoceptor affinity decreased for these compounds, selectivity was not gained over the parent 3-hydroxymethyl compounds (1, 2) due to a loss in PNMT inhibitory potency.